Case
Every successful collaboration is a testament to our quality and capabilities. Learn how we provide reliable screw pumps and accessories to customers worldwide, creating value together.
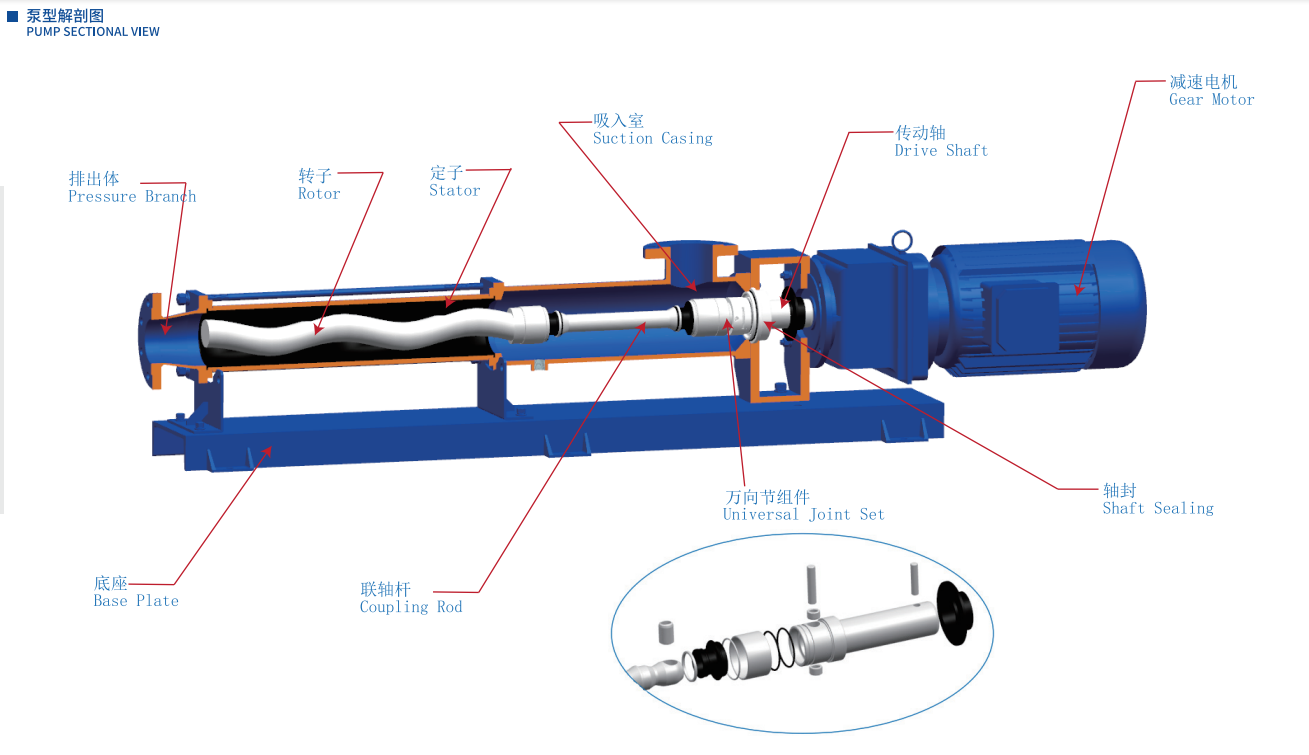
When evaluating hygienic transfer equipment, one core question is: How does food grade screw pump meet FDA standards? For technical assessors, the answer starts with material selection—especially wetted components such as stators, rotors, seals, and elastomers. In food processing, the right FDA-compliant materials directly affect product safety, cleanability, durability, and replacement cost, making material choice the most critical factor in pump qualification.
Why technical assessors should use a material-first checklist
A food grade progressive cavity screw pump is rarely rejected because the pumping principle is unsuitable. More often, qualification is delayed because one or two contact materials are unclear, unsupported, or mismatched to the product being transferred. That is why a checklist-based review is more efficient than starting with general brochures or broad hygienic claims.
For most food applications, the first 30 to 60 minutes of technical assessment should focus on four high-impact areas: all wetted materials, temperature range, cleaning regime, and spare-part traceability. If these points are not aligned early, later discussion about flow rate, pressure, or motor configuration may not solve the actual compliance risk.
This also explains the practical meaning behind the question, How does food grade screw pump meet FDA standards? It is not only about whether a pump is used in food service. It is about whether every product-contact component can support safe transfer under defined process conditions such as 5°C cold dairy service, 25°C sauce transfer, or 80°C CIP exposure.
Start with these priority checks
- Confirm the complete wetted material list, not only the pump housing. Stator elastomer, rotor metallurgy, mechanical seal faces, O-rings, gaskets, and lubricant isolation points all matter.
- Match the material to the medium. Acidic fruit puree, sugar syrup, edible oil, and protein slurry do not stress elastomers in the same way.
- Check operating limits in real ranges, such as viscosity from 1,000 to 100,000 cP, pressure from 2 to 12 bar, or cleaning temperature up to 90°C.
- Review spare-part replacement frequency and procurement lead time, because maintenance cost over 12 to 24 months often outweighs the initial pump price difference.
For assessors comparing domestic and imported-brand replacement components, dimensional consistency and equivalent service life are also practical review items. In many projects, reducing the spare-part sourcing cycle from several weeks to a shorter local supply window can improve uptime and lower inventory pressure without changing the installed pump footprint.
Core checklist: which materials matter most in FDA-related food pump review
When asking How does food grade screw pump meet FDA standards?, technical teams should rank materials by contact risk and failure impact. In progressive cavity screw pumps, not all parts carry equal importance. The highest priority usually goes to elastomer stators and sealing elements because these components combine direct product contact, wear, chemical exposure, and thermal cycling.
The second priority is metallic wetted parts, especially rotors and product-contact metal surfaces. Here, corrosion resistance, surface finish, and compatibility with cleaning chemicals affect both hygiene and service life. A rotor that is dimensionally accurate but poorly matched to the medium can still drive faster stator wear and higher total replacement cost.
The table below gives a practical priority framework for technical assessors reviewing food grade screw pump materials.
This ranking helps avoid a common mistake: overemphasizing the external stainless housing while underchecking the elastomer system. In many food transfer lines running 8 to 20 hours per day, stator and seal materials determine maintenance intervals far more directly than the outer frame or drive type.
Material-by-material review points
Stator elastomer
The stator is often the first component to review because it sees continuous friction, pressure, and product contact. Depending on whether the medium is oily, acidic, sugary, or protein-rich, different elastomer formulations may perform very differently. Even a 10°C to 15°C increase in cleaning temperature can change swelling behavior and shorten service intervals.
Rotor material
The rotor must maintain dimensional accuracy against the stator while resisting corrosion and wear. For technical assessors, the practical question is not only whether the rotor is metallic, but whether its surface and geometry remain stable during repeated food transfer and washdown cycles over 6 to 18 months of use.
Seals, O-rings, and auxiliary contact materials
These smaller parts are easy to overlook, yet they frequently decide pass or fail in hygienic review. If the main wetted path is suitable but a secondary gasket compound is not, the overall compliance confidence weakens. This is one reason the question How does food grade screw pump meet FDA standards? must always be answered at assembly level, not just at housing level.
How to judge material fit by product, process, and cleaning conditions
A material can be suitable in one food line and problematic in another. Technical assessors should therefore build the evaluation around process reality. The same screw pump may handle yogurt at low shear and moderate temperature very well, yet require a different stator compound for hot sugar syrup or fruit concentrate with acidic cleaning cycles.
The practical review should cover at least three operating windows: production transfer, temporary idle condition, and cleaning exposure. For example, a pump may run product at 20°C to 35°C, stand loaded for 2 to 4 hours between batches, and then see 70°C to 90°C cleaning fluid. All three conditions affect the actual life of elastomers and seals.
The table below helps connect food type and process conditions to the most relevant material checks.
This process-based approach provides a more complete answer to How does food grade screw pump meet FDA standards? The answer depends on matching FDA-suitable contact materials with the actual thermal, chemical, and mechanical stress profile of the line. Material compliance on paper is useful, but process fit is what protects long-term reliability.
Recommended checklist before approval
- List all wetted components and identify each material individually, including seals and gaskets.
- Define product characteristics: viscosity range, pH tendency, solids content, oil content, and expected temperature.
- Define cleaning exposure: CIP frequency per day or per week, cleaning chemical type, and maximum cleaning temperature.
- Review expected service interval for stators and rotors, especially under continuous duty or multi-shift operation.
- Confirm replacement-part availability and whether dimensions are fully interchangeable with installed imported-brand units.
For many assessment teams, this five-step sequence reduces uncertainty faster than asking for a broad compliance statement. It also creates a stronger basis for comparing a new pump purchase with a spare-part replacement strategy for existing progressive cavity screw pumps.
Common overlooked risks that affect qualification and lifecycle cost
Even experienced reviewers can miss failure points when time is limited. The most common gap is reviewing nominal material type but not checking the full operating context. A stator suitable for food contact may still fail prematurely if exposed to dry running, frequent start-stop cycles, or cleaning chemistry outside the intended range.
Another frequent issue is treating replacement compatibility as only a dimensional question. For progressive cavity pumps, matching geometry is necessary, but equivalent wear behavior, rotor-stator fit stability, and practical service life are equally important. Over a 12-month maintenance cycle, a cheaper part that fails early can raise labor, downtime, and sanitation verification cost.
Technical assessors should also watch for hidden contamination pathways. These include difficult-to-clean seal areas, non-ideal dead zones, or material transitions where residues may collect after sticky media transfer. In food service lines running daily batch changes, these details can matter more than nameplate flow capacity.
Risk reminders worth checking line by line
- Do not assume all elastomers in one pump assembly are identical. Main stator material and small seal materials may differ.
- Check whether the pump will face intermittent dry running, even for 30 to 90 seconds during tank changeover, because this can sharply increase stator stress.
- Review whether product abrasiveness or particles will accelerate rotor-stator wear and shorten expected maintenance intervals.
- Confirm whether cleaning chemicals and concentration levels are consistent across all plants, especially in multi-site standardization projects.
- Include spare-part sourcing risk in the evaluation. Long lead times can force overstocking and tie up maintenance budget.
A practical cost warning
In many food plants, stator and rotor replacement cost is not limited to parts. It includes sanitation downtime, technician labor, production rescheduling, and post-maintenance restart checks. This is why a technically suitable, dimensionally interchangeable spare-part option with stable quality can deliver meaningful lifecycle value beyond initial purchase price.
Execution guide: what to prepare before supplier discussion
If your team is actively assessing a food grade screw pump or replacement parts, the fastest path is to prepare a compact technical package before discussion. This helps suppliers answer the real question—How does food grade screw pump meet FDA standards?—with application-specific recommendations instead of generic statements.
A useful technical package can usually be built in 1 to 2 working days if process data is available. It should include the medium name, viscosity range, target flow, discharge pressure, operating temperature, CIP temperature, run schedule, and installed pump model or spare-part dimensions. With this information, assessors can compare material options much more accurately.
For plants already using imported-brand progressive cavity screw pumps, spare-part replacement strategy should be discussed as part of qualification. If locally manufactured stators and rotors can fully replace imported-brand parts with consistent dimensions and equivalent service life, this may shorten procurement cycles and reduce future maintenance cost without requiring major system redesign.
What to send for faster technical review
With these inputs, the supplier can discuss material selection, replacement compatibility, expected wear points, and delivery planning with much greater accuracy. This is especially useful when balancing hygienic requirements with a cost-effective spare-parts strategy.
Why choose us for screw pump and spare-part evaluation
Shanghai Huanyi Machinery Co., Ltd., founded in 2014, focuses on the production of imported-brand progressive cavity screw pumps and spare parts, with particular experience in single screw pumps and accessories. For technical assessors, this means discussions can stay close to the real issues: wetted materials, interchangeability, service life, maintenance cycle, and total spare-part cost.
Our manufactured stators and rotors can fully replace those of imported screw pump brands, with consistent dimensions and equivalent service life compared with original imported parts. For plants under pressure to reduce lead time, control maintenance budgets, or avoid prolonged spare-part procurement cycles, this can be a practical option worth reviewing.
If you are evaluating How does food grade screw pump meet FDA standards?, contact us with your pump model, process medium, temperature range, cleaning conditions, and spare-part requirements. We can support parameter confirmation, material review, product selection, delivery cycle discussion, interchangeable stator and rotor planning, and quotation communication based on your actual application.
Send Us A Message
